Plasma Medical Aesthetic Device RK3568 ARM+FPGA Solution
How Plasma Aesthetic Devices Work
Plasma medical aesthetic devices achieve skin rejuvenation through a precise photochemical mechanism: a high-voltage, low-current electrical field ionizes atmospheric nitrogen and oxygen at the skin surface, creating a plasma arc. The free radicals and reactive oxygen species generated by this arc penetrate into the dermis, triggering a controlled inflammatory response. That response stimulates fibroblasts to synthesize new collagen and elastin, gradually restoring the skin's structural support network and reducing the appearance of fine lines and wrinkles — without ablating the epidermis the way traditional laser resurfacing does.
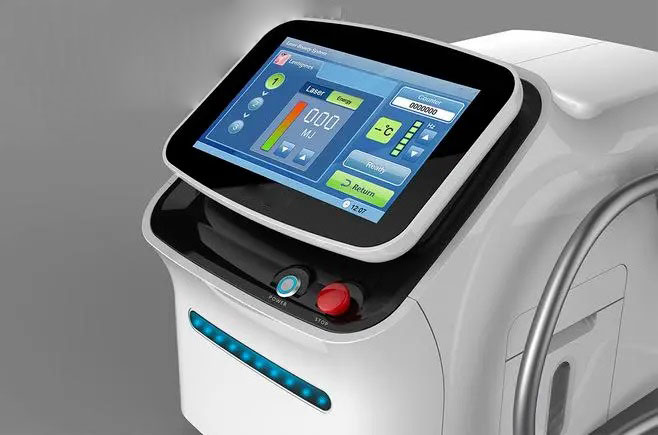
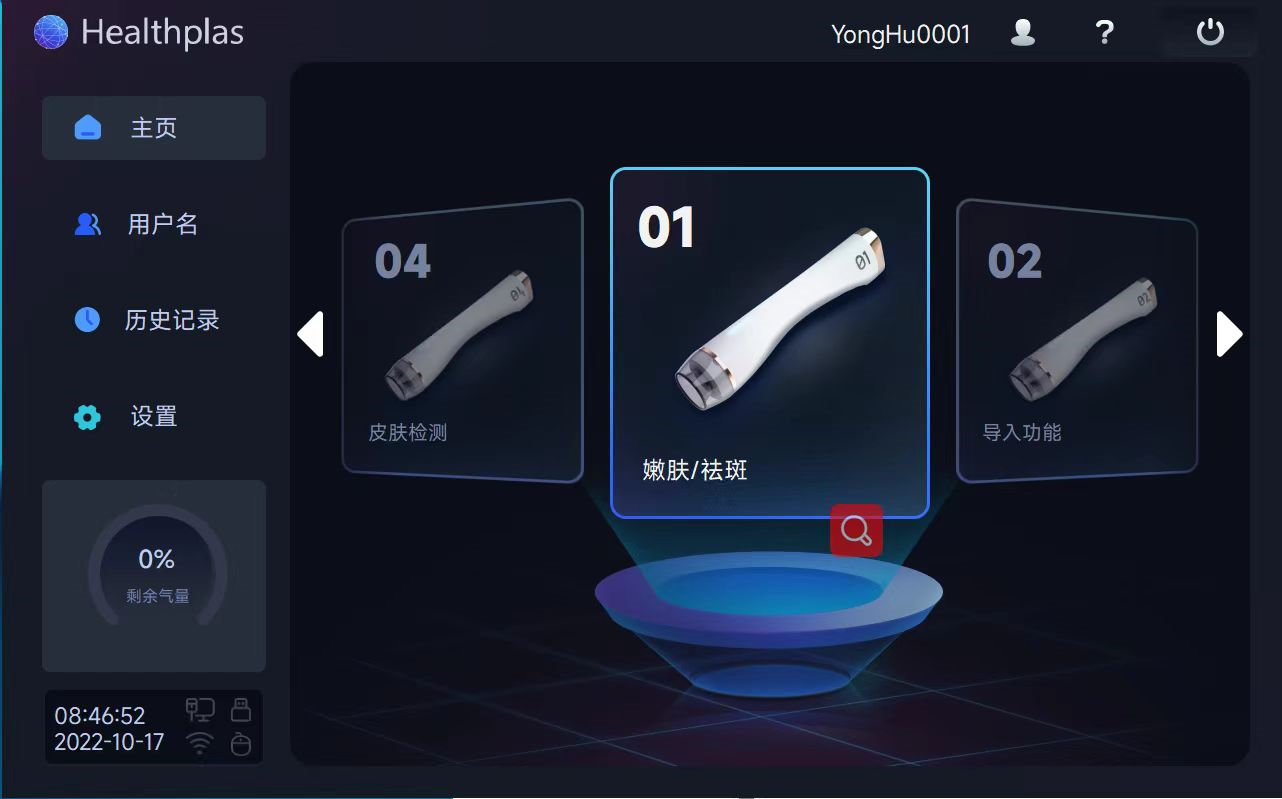
The clinical control system behind a device like this carries its own engineering challenges. The handpiece must deliver repeatable, calibrated energy pulses while the host controller simultaneously manages a live video feed for procedural guidance, a touchscreen UI for clinician interaction, treatment parameter logging, and safety interlocks — all in a medically certified enclosure.
The RK3568 ARM + FPGA Hardware Architecture
Sienovo's solution centers on the Rockchip RK3568, a domestically designed (国产化) SoC that pairs a quad-core ARM Cortex-A55 cluster with a dedicated NPU and a capable Mali-G52 GPU. For a medical aesthetic controller, the key attributes are:
- Video processing headroom. The RK3568 includes a hardware ISP, H.264/H.265 encode/decode engines, and multiple display output paths. This allows the system to handle real-time camera feeds and rich GUI rendering without CPU-bound bottlenecks.
- Long-term supply commitment. Industrial and medical customers cannot tolerate mid-lifecycle part discontinuations. Rockchip's extended supply roadmap for the RK3568 directly addresses this requirement.
- Peripheral richness. USB 3.0, PCIe 2.0, MIPI CSI/DSI, and dual Gigabit Ethernet give integrators flexible options for peripherals, external storage, and network connectivity.
The FPGA in the ARM+FPGA pairing handles the real-time, deterministic side of the workload: precise pulse-width modulation of the high-voltage driver circuitry, microsecond-level safety monitoring (overcurrent, arc duration limits, handpiece temperature), and hardware interlocks that operate independently of the Linux kernel running on the ARM cores. This hard separation between the application processor and the safety-critical FPGA is a well-established pattern in medical device design — it ensures that a kernel panic or a UI thread deadlock cannot disable protective shutdown logic.
Qt-Based Cross-Platform UI
The host application runs under Linux on the RK3568 and is built with the Qt framework. Qt's value in this context comes from several directions:
- Rich widget and QML ecosystem. Clinician-facing UIs for aesthetic devices require smooth touch interaction, animated parameter controls, and clear data visualization. Qt's GPU-accelerated rendering pipeline (via OpenGL ES on the Mali GPU) delivers this without requiring a web browser stack.
- Cross-platform portability. Development and testing can happen on x86 Linux or Windows workstations, then the same codebase deploys to the ARM target with a recompile. This dramatically shortens iteration cycles during device validation.
- Mature medical-UI precedent. Qt has a long history in medical imaging and point-of-care terminals, so its threading model, signal/slot architecture, and serialport/USB libraries are well understood by teams building IEC 62304-aware software.
Solution Topology
The block diagram below shows how the major subsystems connect — the RK3568 SoC as the central application processor, the FPGA handling real-time pulse control and safety logic, the display and camera interfaces, and the high-voltage plasma generation circuitry.
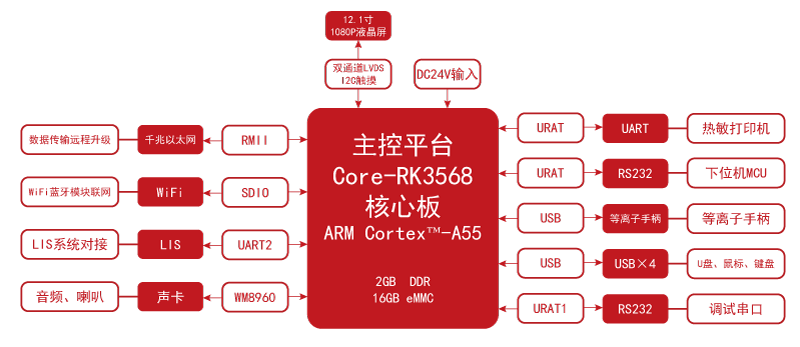
Why This Architecture Fits Medical Aesthetics
Combining a high-performance applications processor with a hard-real-time FPGA, rather than relying on a microcontroller alone, gives the platform room to grow: future iterations can add AI-assisted skin analysis (leveraging the RK3568's NPU), video recording for procedure documentation, or cloud connectivity for treatment data — without redesigning the safety-critical FPGA layer. The use of a domestically sourced SoC also simplifies supply chain certification in markets where regulatory bodies require component traceability, a concern increasingly relevant in the medical device industry.